 If the implanted pulse generator (IPG) or the electrodes are within or near the field of view, some image degradation (distortion, artifact, etc.) is to be expected.Ī new technology was launched recently in both Europe and later in the United States, with which, according to the approval granted by the regulatory authorities, patients can safely undergo a full-body MRI scan under specific conditions. 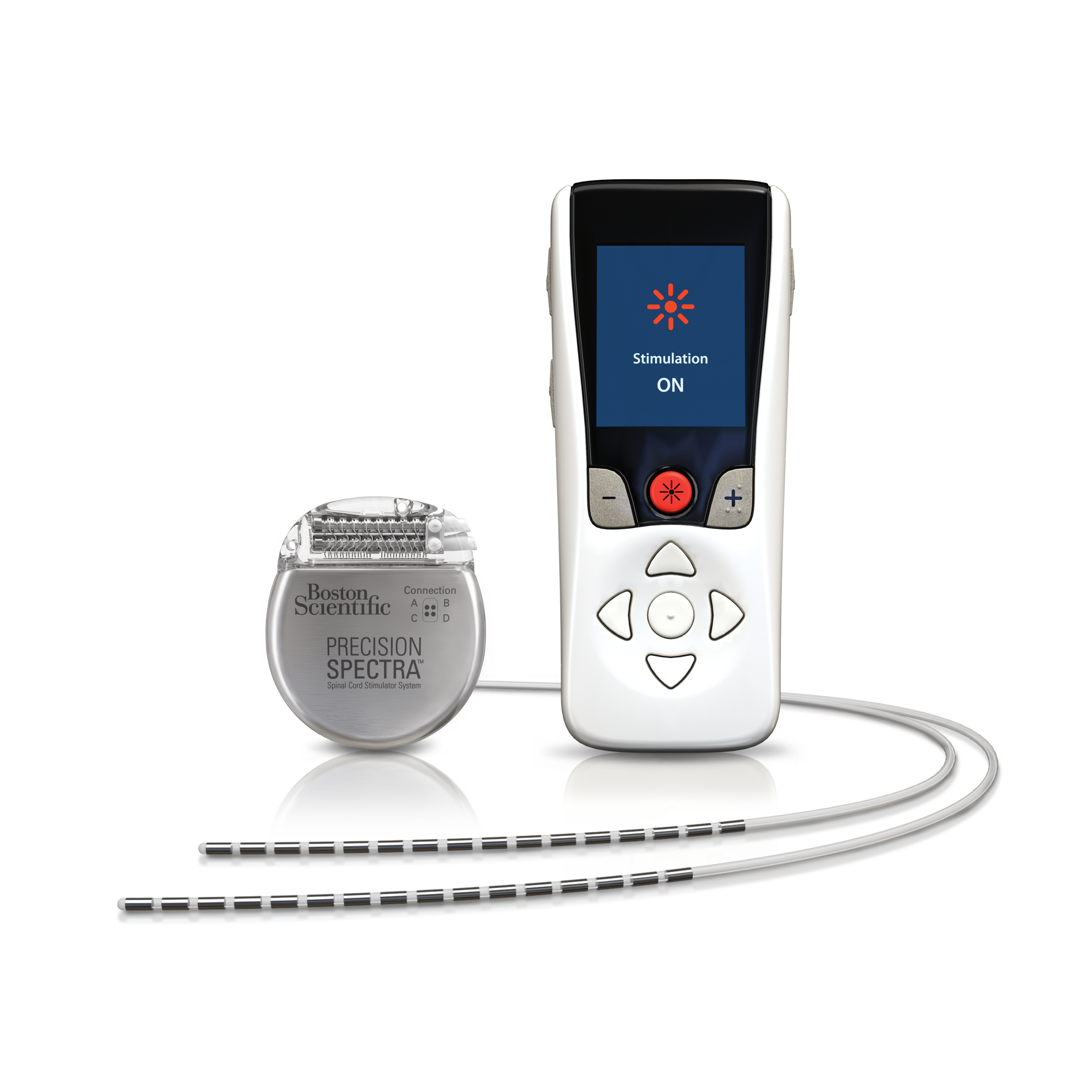 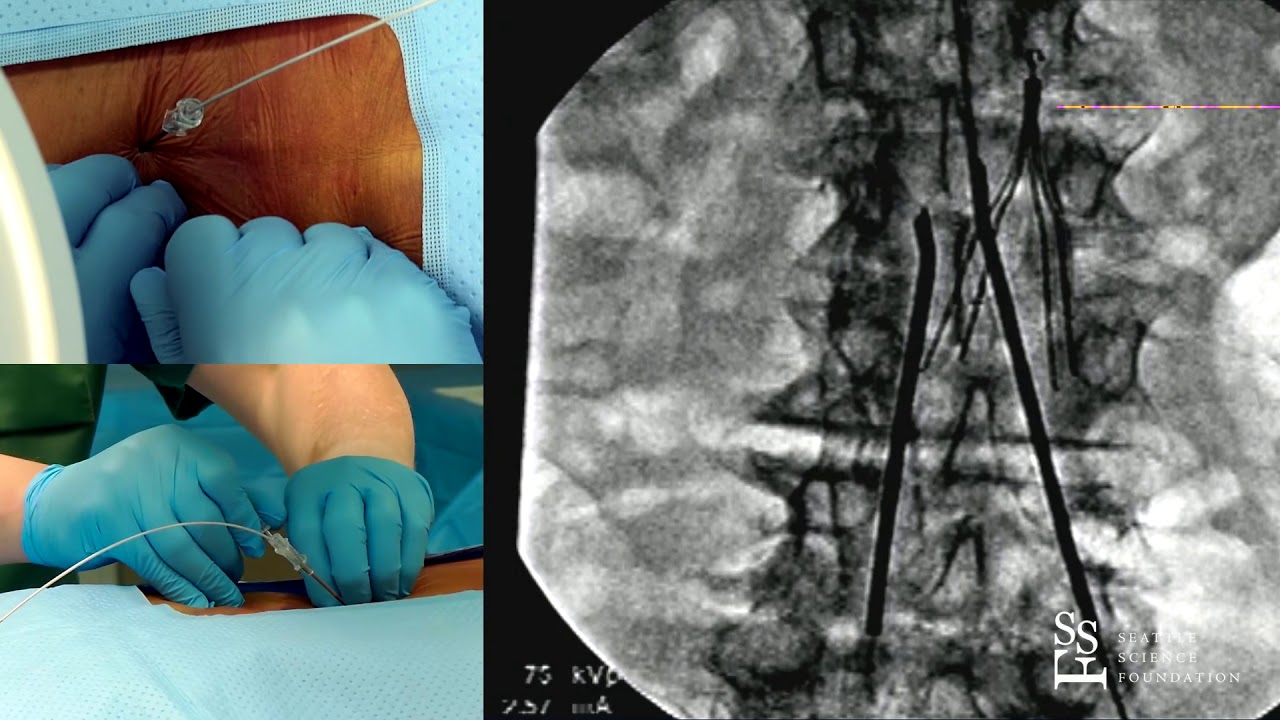 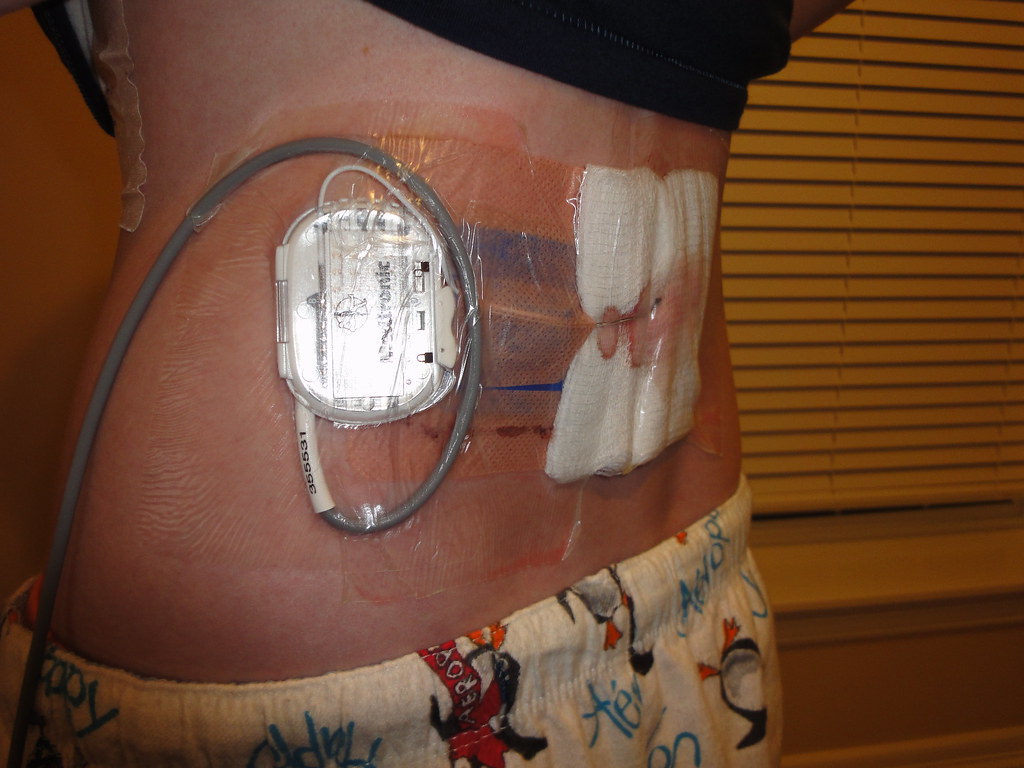
Less concerning but also important are the effects of the medical devices on the operation of the MRI scanner, resulting in poor-quality images due to excessive artifacts, possibly because of the RF currents induced on the surface of the lead. These can result in an interaction with the medical device, that is, rotational force (torque) on an object with tearing of tissues rotation of an object for alignment with the field and translational force on an object resulting in (1) tearing of tissues, (2) acceleration of an object into the bore of the magnet (“missile effect”), (3) induced currents due to the rate of change of the magnetic flux density with time (T/s) causing device malfunction or failure, and (4) RF-induced currents resulting in device heating and patient burns (thermal and electrical) (Table 0002). The main concern about performing MRI with a SCS in situ relates to one or a combination of the four main components that make up the MRI environment: the static magnetic field, the static magnetic field spatial gradient, the gradient magnetic field, and the RF field. Therefore, in general, the presence of this object is considered to be a contraindication for an MR procedure and/or for an individual to enter the MR environment depending on the nature of the object or item. Other risks or a different hazard may also exist. Unsafe 1: The object is considered to pose a potential or realistic risk or hazard to a patient or individual in the MR environment (static magnetic field strength of 1.5 T) primarily as the result of movement or displacement of the object. Frequently, this information is best found at the company's website. Consult the manufacturer of the particular device for the latest safety information. Please refer to the specific criteria for performing the MR procedure by reviewing the information for the object on this website. 
The object may or may not be safe for the patient undergoing an MR procedure or an individual in the MR environment, depending on the specific conditions that are present.Ĭonditional 5: This object is acceptable for a patient undergoing an MR procedure or an individual in the MR environment only if specific guidelines or recommendations are followed (see specific information for a given object on this website and contact the manufacturer for further information). The MR conditions in which the device was tested should be specified in conjunction with the term MR compatible as a device that is safe under one set of conditions may not be found to be so under more extreme MR conditions. The device, when used in the MR environment, has been demonstrated to present no additional risk to the patient or other individual, but may affect the quality of the diagnostic information.Ī device is considered MR compatible if it is MR safe and if it, when used in the MR environment, has been demonstrated to neither significantly affect the quality of the diagnostic information nor have its operations affected by the MR device. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
